
🏭 Industry: Pharmaceutical Manufacturing (OSD & Regulated Markets)
💼 Job Type: Full-Time | Quality Control & Quality Assurance
📍 Location: Pithampur (Indore), Madhya Pradesh
About Piramal Pharma Limited
Piramal Pharma Limited is a globally recognized pharmaceutical manufacturing and services organization with a strong presence across regulated markets. Known for its robust quality systems, advanced analytical capabilities, and compliance-driven culture, Piramal offers professionals an excellent platform for long-term career growth in QC, QA, and validation domains.
To strengthen its Pithampur (Indore) manufacturing facility, Piramal Pharma is conducting a Walk-In Interview in February 2026 for experienced professionals and freshers in Quality Control (QC) and Quality Assurance (QA).
Current Job Openings – Department Wise
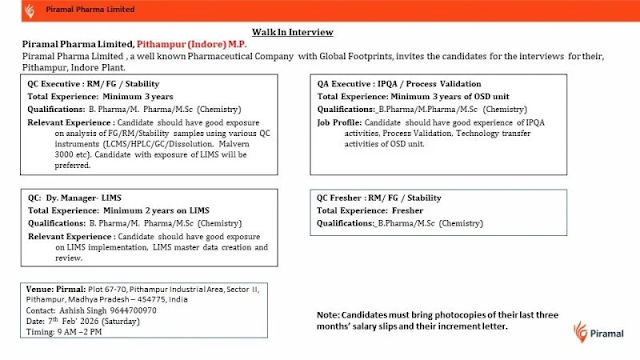
1️⃣ Quality Control (QC) – Executive (RM / FG / Stability)
Experience: Minimum 3 Years
Qualification: B.Pharm / M.Pharm / M.Sc (Chemistry)
Location: Pithampur (Indore), MP
Role Highlights
- Raw Material, Finished Goods & Stability analysis
- Handling instruments: HPLC, GC, LC-MS, Malvern, Dissolution
- GMP-compliant testing & documentation
- LIMS exposure preferred
2️⃣ Quality Assurance (QA) – Executive (IPQA / Process Validation)
Experience: Minimum 3 Years (OSD Unit)
Qualification: B.Pharm / M.Pharm / M.Sc (Chemistry)
Location: Pithampur (Indore), MP
Role Highlights
- IPQA activities & shop-floor compliance
- Process validation & technology transfer
- QA operations in OSD manufacturing
- Regulatory & GMP documentation
3️⃣ Quality Control (QC) – Deputy Manager (LIMS)
Experience: Minimum 2 Years on LIMS
Qualification: B.Pharm / M.Pharm / M.Sc (Chemistry)
Location: Pithampur (Indore), MP
Role Highlights
- LIMS implementation & master data management
- Compliance review & system validation
- Regulated pharma environment exposure
4️⃣ Quality Control (QC) – Fresher (RM / FG / Stability)
Experience: Fresher
Qualification: B.Pharm / M.Sc (Chemistry)
Location: Pithampur (Indore), MP
Role Highlights
- Entry-level QC testing exposure
- Training on GMP, documentation & lab practices
- Ideal start for a pharma QC career
Walk-In Interview Details
📅 Date: 08 February 2026 (Saturday)
⏰ Time: 9:00 AM to 2:00 PM
📍 Venue:
Piramal Pharma Limited
Plot No. 67–70, Pithampur Industrial Area, Sector-II,
Pithampur, Madhya Pradesh – 454775
Documents to Carry
- Updated Resume
- Last 3 months’ salary slips (for experienced candidates)
- Last increment letter
- Educational certificates & ID proof
Who Can Apply
- B.Pharm / M.Pharm / M.Sc (Chemistry) candidates
- Experienced professionals in QC, QA, IPQA, Validation & LIMS
- Freshers seeking QC entry-level roles
- Candidates with OSD & GMP exposure
Why Join Piramal Pharma
- Work with a global pharmaceutical leader
- Exposure to regulated markets & advanced quality systems
- Strong career growth in QC, QA & validation
- Hands-on experience with modern analytical instruments
- Stable, compliance-driven work culture
Interview Preparation Tips
- Revise GMP, IPQA, QC & validation fundamentals
- Be confident about instrument handling & documentation
- Prepare real examples from audits, deviations, or investigations
- Carry all documents neatly arranged
Frequently Asked Questions (FAQs)
Q1. Is this Piramal Pharma walk-in open for freshers?
Yes, QC Fresher positions are open for eligible candidates.
Q2. What qualifications are required?
B.Pharm, M.Pharm, or M.Sc (Chemistry).
Q3. Is this a walk-in or online interview?
This is a direct walk-in interview.
Q4. What is the job location?
Pithampur (Indore), Madhya Pradesh.
Q5. Is experience mandatory for all roles?
No. Experience is required for Executive & Manager roles, while QC Fresher roles are open to freshers.
Q6. What documents should I bring?
Resume, salary slips (if applicable), increment letter, and certificates.
Q7. Are OSD unit candidates eligible?
Yes, especially for QA IPQA & Process Validation roles.
Piramal Pharma Limited Walk-In Interview 2026 for QC & QA Professionals

